Pharmaceutical Membrane Filtration Market Revenue, Trends, and Strategic Insights by 2035
Table of Contents
TogglePharmaceutical Membrane Filtration Market Size
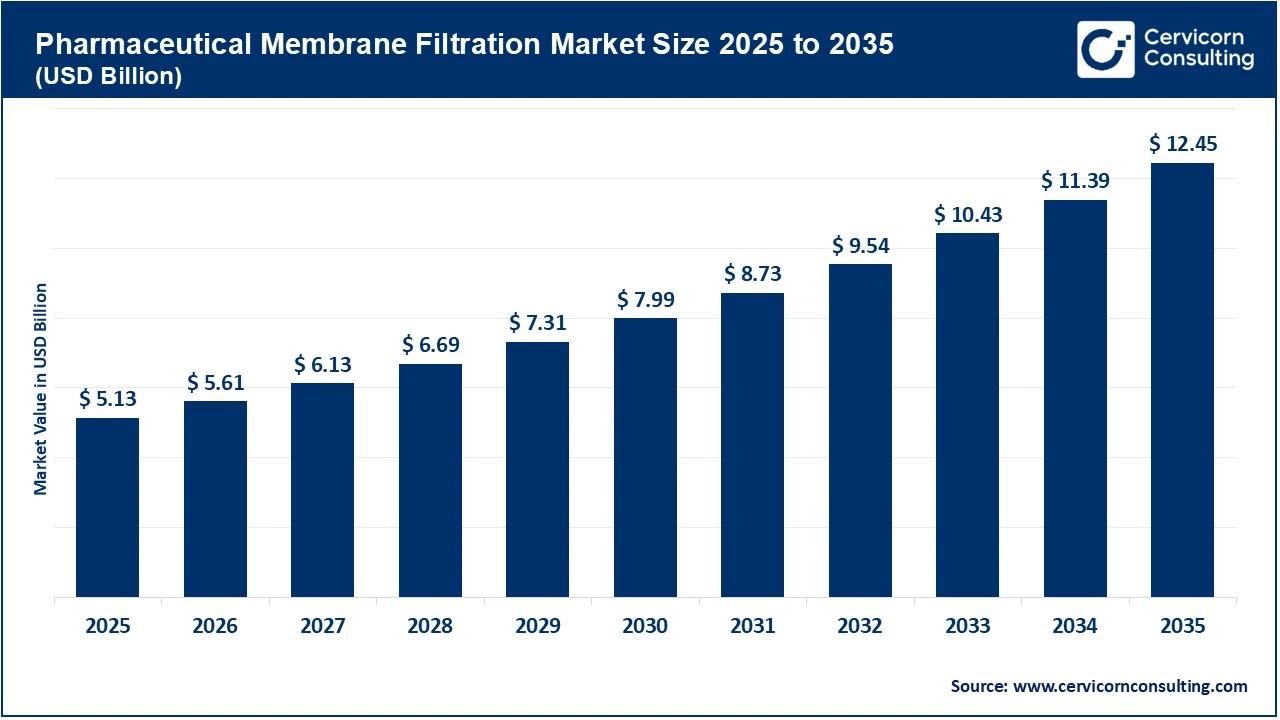
The global pharmaceutical membrane filtration market reached an estimated valuation of USD 5.13 billion in 2025 and is projected to grow substantially, attaining approximately USD 12.45 billion by 2035. This growth reflects a strong compound annual growth rate (CAGR) of around 9.3% throughout the forecast period.
What is the Pharmaceutical Membrane Filtration Market?
The pharmaceutical membrane filtration market refers to the industry focused on the development, manufacturing, and application of membrane-based filtration technologies used in pharmaceutical and biopharmaceutical production processes. These membranes are semi-permeable barriers that selectively allow molecules or particles to pass through based on size, charge, or chemical properties. They are widely used in processes such as sterilization, clarification, separation, purification, and concentration of pharmaceutical products.
Membrane filtration technologies include microfiltration, ultrafiltration, nanofiltration, and reverse osmosis, each serving specific roles in drug manufacturing and bioprocessing. These systems are critical in ensuring that pharmaceutical products meet stringent regulatory standards for sterility, purity, and safety.
As pharmaceutical production becomes more complex with biologics, vaccines, and gene therapies, membrane filtration has become an indispensable part of modern drug manufacturing infrastructure.
Why is Pharmaceutical Membrane Filtration Important?
Pharmaceutical membrane filtration is vital because it ensures product safety, regulatory compliance, and manufacturing efficiency. In drug production, even microscopic contamination can compromise patient safety, making filtration a non-negotiable step.
Key reasons for its importance include:
- Ensures sterility and removal of microorganisms
- Supports production of biologics, vaccines, and injectables
- Helps meet FDA, EMA, and GMP compliance standards
- Enables high-purity separation of proteins and biomolecules
- Reduces contamination risk in aseptic processing
- Improves manufacturing efficiency and yield
- Supports single-use and continuous manufacturing systems
With rising demand for advanced therapeutics and biologics, membrane filtration plays a central role in maintaining quality assurance in pharmaceutical manufacturing pipelines.
Pharmaceutical Membrane Filtration Market Growth Factors
The pharmaceutical membrane filtration market is experiencing strong growth due to increasing biologics production, rising demand for sterile injectables, expansion of vaccine manufacturing, stringent regulatory standards, adoption of single-use technologies, growth in contract manufacturing organizations (CMOs), rising investment in biopharmaceutical R&D, technological advancements in membrane materials, expansion of pharmaceutical production in emerging markets, and increasing need for high-efficiency purification and separation systems in complex drug development processes, which collectively are driving sustained global market expansion across both developed and developing regions.
Get a Free Sample: https://www.cervicornconsulting.com/sample/2935
Leading Companies in the Pharmaceutical Membrane Filtration Market
1. Merck KGaA
- Specialization: Life science tools, filtration membranes, and bioprocess solutions
- Key Focus Areas: Microfiltration, ultrafiltration, sterile filtration systems
- Notable Features: Strong R&D in biomaterials, advanced single-use filtration systems
- 2024 Revenue: ~€21–22 billion (company-wide estimate)
- Market Share: One of the global leaders in life sciences filtration segment
- Global Presence: Strong across Europe, North America, and Asia-Pacific
Merck KGaA is a dominant player offering advanced filtration technologies used in biologics and sterile drug manufacturing.
2. Danaher Corporation
- Specialization: Biotechnology tools through subsidiaries like Cytiva
- Key Focus Areas: Bioprocess filtration, downstream purification systems
- Notable Features: Strong integration of filtration with bioprocessing workflows
- 2024 Revenue: ~$23–25 billion (life sciences segment-driven)
- Market Share: Among top 3 global players in filtration technologies
- Global Presence: North America, Europe, China, India, Japan
Danaher benefits from Cytiva’s strong presence in bioprocess filtration, making it a key innovator in biologics manufacturing systems.
3. Sartorius AG
- Specialization: Bioprocessing and laboratory filtration solutions
- Key Focus Areas: Sterile filtration, membrane systems for cell culture
- Notable Features: Strong focus on single-use technologies
- 2024 Revenue: ~$3.4–3.7 billion
- Market Share: Leading in biopharma filtration consumables
- Global Presence: Europe, U.S., Asia-Pacific
Sartorius is known for its advanced filtration membranes tailored for biotech and pharmaceutical manufacturing.
4. Thermo Fisher Scientific
- Specialization: Life sciences solutions and biopharma services
- Key Focus Areas: Sterile filtration, purification, laboratory filtration systems
- Notable Features: Strong acquisition-driven expansion strategy
- 2024 Revenue: ~$42+ billion
- Market Share: One of the largest players in biopharma tools and filtration
- Global Presence: Global (especially strong in U.S., China, and Europe)
Thermo Fisher has significantly expanded its filtration capabilities through strategic acquisitions, strengthening its position in drug manufacturing solutions.
5. Parker Hannifin Corporation
- Specialization: Motion and control technologies, industrial filtration
- Key Focus Areas: High-purity filtration, gas-liquid separation systems
- Notable Features: Engineering-driven filtration solutions for pharma processes
- 2024 Revenue: ~$20+ billion (company-wide estimate)
- Market Share: Strong niche player in process filtration systems
- Global Presence: North America, Europe, Asia
Parker Hannifin provides robust filtration systems used in upstream and downstream pharmaceutical processes, ensuring process reliability.
Leading Trends and Their Impact on the Market
The pharmaceutical membrane filtration market is evolving rapidly due to several transformative trends:
1. Single-Use Filtration Systems
Single-use technologies reduce contamination risk, eliminate cleaning validation, and improve operational flexibility. This trend is significantly boosting demand across biopharmaceutical manufacturing facilities.
2. Growth of Biologics and Cell & Gene Therapy
The rise of complex biologics requires advanced filtration solutions for purification and separation, driving innovation in membrane technologies.
3. Continuous Manufacturing Adoption
Continuous production processes require robust filtration systems that operate consistently, increasing demand for integrated membrane solutions.
4. Smart Filtration Systems (Industry 4.0 Integration)
Digital monitoring, predictive maintenance, and real-time analytics are improving efficiency and reducing downtime in filtration operations.
5. High-Performance Membrane Materials
Advancements in polymer science and nanotechnology are improving membrane durability, selectivity, and efficiency.
6. Expansion in Emerging Markets
Countries in Asia-Pacific are rapidly expanding pharmaceutical manufacturing, increasing adoption of advanced filtration systems.
Successful Examples of Pharmaceutical Membrane Filtration Applications Worldwide
- COVID-19 Vaccine Production (Global): Membrane filtration was critical in purifying mRNA vaccine components and ensuring sterile injectable formulations.
- Monoclonal Antibody Manufacturing (USA & Europe): Companies use ultrafiltration systems to concentrate and purify antibodies for cancer treatment.
- Biologic Drug Manufacturing in India: Large-scale CMOs use membrane filtration for cost-efficient production of biosimilars.
- Gene Therapy Production (Switzerland & USA): High-precision filtration ensures removal of viral contaminants during vector production.
- Water Purification in Pharma Plants (Global): Reverse osmosis membranes ensure ultra-pure water for injectable drug production.
These applications demonstrate how membrane filtration supports modern pharmaceutical innovation across therapeutic areas.
Global Regional Analysis
North America
North America dominates the pharmaceutical membrane filtration market due to advanced biopharmaceutical infrastructure, high R&D spending, and strong regulatory frameworks. The U.S. leads in biologics and vaccine production, driving demand for advanced filtration systems.
Government agencies like the FDA enforce strict GMP and sterility requirements, pushing pharmaceutical companies to adopt high-performance filtration technologies.
Europe
Europe is a major hub for pharmaceutical manufacturing, especially in Germany, Switzerland, and the UK. The region benefits from strong regulatory standards under the EMA, encouraging adoption of advanced filtration systems and sustainable manufacturing practices.
EU initiatives promoting biopharmaceutical innovation and clean manufacturing processes are further supporting market growth.
Asia-Pacific
Asia-Pacific is the fastest-growing region in the pharmaceutical membrane filtration market. Countries such as China and India are expanding pharmaceutical production capacities significantly.
Government initiatives supporting local drug manufacturing, vaccine production, and biosimilars are accelerating demand for membrane filtration technologies.
Latin America & Middle East & Africa
These regions are witnessing steady growth due to increasing healthcare investments, expansion of pharmaceutical production facilities, and improving regulatory frameworks.
Governments are focusing on strengthening local drug manufacturing capabilities, which is indirectly boosting demand for filtration technologies.
Government Initiatives and Policies Shaping the Market
- FDA (U.S.) regulations enforcing strict sterility and filtration validation standards
- EMA guidelines (Europe) promoting high-quality biologics manufacturing
- China’s pharmaceutical innovation policies supporting domestic biotech expansion
- India’s “Make in India” initiative encouraging pharmaceutical manufacturing growth
- WHO guidelines for vaccine production influencing global filtration standards
- Good Manufacturing Practices (GMP) globally standardizing filtration requirements
These regulatory frameworks ensure high adoption of membrane filtration systems across pharmaceutical production facilities worldwide.
Final Insight
The pharmaceutical membrane filtration market is a rapidly evolving sector driven by biologics expansion, technological advancements, and strict regulatory compliance requirements. With major global companies investing heavily in innovation and emerging markets expanding production capacity, membrane filtration is becoming a foundational technology in modern pharmaceutical manufacturing.
To Get Detailed Overview, Contact Us: https://www.cervicornconsulting.com/contact-us
Read Report: Cell Culture Media Market Revenue, Trends, and Strategic Insights by 2035