Robotics in Cell Therapy Manufacturing Market Size
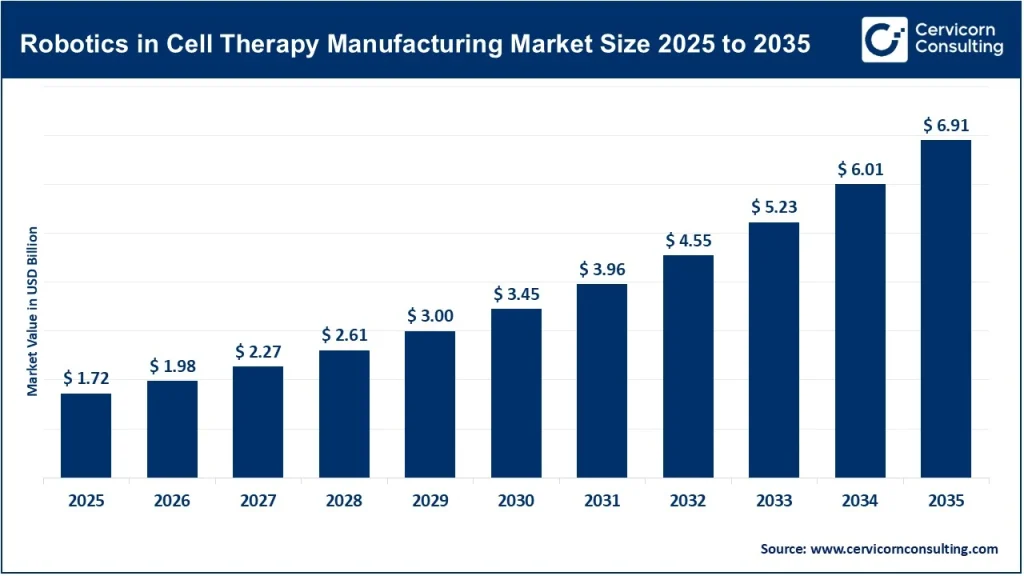
The global robotics in cell therapy manufacturing market is expected to grow from approximately USD 1.72 billion in 2025 to about USD 6.91 billion by 2035, registering a CAGR of around 14.9% during the forecast period.
What is the Robotics in Cell Therapy Manufacturing Market?
The robotics in cell therapy manufacturing market refers to the global ecosystem of automated hardware platforms, digital control systems, AI-enabled analytics solutions, and closed-loop production technologies designed to support the large-scale manufacturing of therapeutic cells. These robotic platforms automate key workflow stages including cell isolation, activation, genetic modification, expansion, harvesting, fill-finish operations, and quality control testing.
Traditional cell therapy manufacturing relies heavily on manual laboratory handling steps—often between 50 and 100 process interactions per batch—which introduces variability and contamination risks. Robotic systems reduce manual intervention while improving sterility, traceability, and reproducibility. Fully automated closed systems are increasingly becoming the preferred production architecture because they enable scalable, GMP-compliant workflows aligned with regulatory requirements for clinical and commercial supply.
Modern robotics platforms are also integrating artificial intelligence and digital twins into manufacturing execution systems, enabling predictive maintenance, batch optimization, and process standardization across decentralized facilities. These capabilities are accelerating the transition from small-batch academic production to industrial-scale therapeutic cell manufacturing infrastructure worldwide.
Why Robotics in Cell Therapy Manufacturing is Important
Robotics plays a transformative role in enabling the commercialization of cell therapies by addressing the most significant bottlenecks limiting global adoption.
First, automation improves sterility and reduces contamination risk. Since cell therapies involve living biological material, even minor deviations in process control can affect therapeutic outcomes. Closed robotic systems maintain standardized conditions throughout production.
Second, robotics increases scalability. Personalized therapies historically required labor-intensive manufacturing models unsuitable for large patient populations. Automated platforms now support parallel batch production across multiple treatment lines.
Third, robotics enhances reproducibility and regulatory compliance. Regulatory agencies increasingly favor automated manufacturing architectures because they improve traceability and batch consistency.
Fourth, automation reduces production costs over time. Robotic workflows minimize labor dependency and facility footprint requirements while improving throughput efficiency.
Finally, robotics enables decentralized manufacturing models. Hospitals and regional treatment centers are increasingly adopting modular robotic platforms capable of producing therapies closer to patients, reducing logistics complexity and treatment turnaround time.
Together, these advantages position robotics as a foundational technology supporting the global expansion of personalized medicine and advanced biologics manufacturing.
Get a Free Sample: https://www.cervicornconsulting.com/sample/2932
Robotics in Cell Therapy Manufacturing Market Growth Factors
The robotics in cell therapy manufacturing market is expanding rapidly due to increasing regulatory approvals of advanced therapies, rising global demand for CAR-T and stem-cell treatments, growing investment in GMP-compliant automation platforms, and the need to reduce contamination risks associated with manual workflows; integration of artificial intelligence and predictive analytics into robotic systems is further improving production efficiency and reducing costs by up to 30% while accelerating research timelines by nearly 44%, encouraging pharmaceutical companies to transition toward closed automated systems; additional growth is supported by expansion of contract manufacturing organizations (CMOs), increasing partnerships between robotics developers and bioprocess equipment providers, adoption of decentralized manufacturing architectures, and strong government funding initiatives promoting advanced therapy commercialization worldwide.
Company Landscape: Key Players Driving Robotics in Cell Therapy Manufacturing
Below is a structured comparison of leading companies shaping the robotics in cell therapy manufacturing ecosystem.
Multiply Labs
Company Specialization: Modular robotic automation for cell therapy production
Key Focus Areas: Closed-system automation, robotic cell-processing modules, GMP integration
Notable Features: Flexible robotic platform architecture compatible with existing bioprocess tools; collaborations with major biopharma equipment providers
2024 Revenue: Estimated privately held (not publicly disclosed)
Market Share: Emerging automation innovator segment
Global Presence: United States, Europe partnerships, global pharma collaborations
Multiply Labs focuses on robotic automation modules designed to integrate seamlessly into existing cell therapy production workflows. Its platforms support scalable manufacturing infrastructure across research and commercial environments.
Cellular Origins
Company Specialization: Robotic manufacturing platforms for scalable CGT production
Key Focus Areas: High-throughput automation, decentralized manufacturing infrastructure
Notable Features: Constellation® platform designed to reduce labor requirements by up to 16× and improve facility space utilization dramatically
2024 Revenue: Privately held
Market Share: Early-stage high-growth automation provider
Global Presence: UK headquarters with global partnerships across North America and Europe
Cellular Origins is developing robotic orchestration platforms designed to industrialize cell therapy manufacturing by enabling large-scale production networks.
Cellares
Company Specialization: End-to-end automated cell therapy manufacturing systems
Key Focus Areas: Integrated robotic production units, closed-system automation, high-throughput batch manufacturing
Notable Features: Cell Shuttle™ platform enables simultaneous production of multiple therapy batches with automated quality control
2024 Revenue: Privately held
Market Share: Leading advanced automation innovator
Global Presence: United States, Europe, Japan manufacturing expansion plans
Cellares is one of the most advanced robotics platform providers aiming to standardize large-scale therapeutic cell production.
Thermo Fisher Scientific
Company Specialization: Bioprocessing equipment and integrated automation technologies
Key Focus Areas: GMP manufacturing tools, cell processing platforms, reagent systems
Notable Features: Integration of robotics with Gibco CTS™ cell therapy manufacturing platforms
2024 Revenue: Over USD 42 billion (corporate revenue across segments)
Market Share: Major global life sciences infrastructure provider
Global Presence: Operations across North America, Europe, Asia-Pacific
Thermo Fisher Scientific provides enabling technologies that integrate robotics into commercial-scale cell therapy workflows.
Cytiva
Company Specialization: Bioprocessing automation and digital manufacturing solutions
Key Focus Areas: Closed-system manufacturing platforms, process analytics, scalable biologics infrastructure
Notable Features: Automation-enabled flexible manufacturing architecture supporting clinical-to-commercial transitions
2024 Revenue: Estimated multi-billion-dollar life sciences segment contribution under parent organization
Market Share: Strong presence in advanced therapy manufacturing solutions
Global Presence: Global operations across major biopharma manufacturing hubs
Cytiva delivers integrated automation platforms that support continuous and modular cell therapy production environments.
Leading Trends Shaping the Robotics in Cell Therapy Manufacturing Market and Their Impact
Shift Toward Fully Automated Closed Systems
Closed robotic manufacturing systems are becoming the dominant production architecture because they improve sterility and compliance while reducing manual handling requirements. These platforms support end-to-end automation from cell isolation to final packaging.
Impact: Enables scalable commercial production and supports regulatory approval pathways.
Integration of Artificial Intelligence in Manufacturing Workflows
AI-enabled robotics platforms now support predictive maintenance, yield optimization, anomaly detection, and automated process adjustments.
Impact: Improves manufacturing reliability while reducing production costs and downtime.
Decentralized Manufacturing Models
Robotic micro-factories located near hospitals are enabling localized therapy production.
Impact: Reduces logistics costs and treatment turnaround time for personalized therapies.
Modular Manufacturing Infrastructure
Plug-and-play robotic modules allow flexible facility scaling without major capital redesign.
Impact: Accelerates adoption among contract manufacturing organizations and research institutions.
Increasing Partnerships Between Robotics Providers and Biopharma Companies
Collaborations such as Multiply Labs–Thermo Fisher integrations highlight ecosystem-level automation development.
Impact: Reduces implementation complexity and improves interoperability between platforms.
Successful Global Examples of Robotics in Cell Therapy Manufacturing
Cellares Cell Shuttle™ Automated Manufacturing Platform
The Cell Shuttle system integrates multiple laboratory instruments into a single robotic unit capable of producing therapies for multiple patients simultaneously while reducing labor requirements by up to 90%. It also enables continuous manufacturing cycles without manual interruptions.
Impact: Demonstrates feasibility of commercial-scale robotic production facilities capable of producing thousands of therapies annually.
Cellular Origins Constellation® Platform
Constellation enables orchestration of multiple automated processing modules within GMP-compliant environments while dramatically improving facility space utilization.
Impact: Supports industrial-scale distributed manufacturing networks.
Multiply Labs Robotic Cell Processing Modules
Multiply Labs collaborates with global bioprocess providers to automate early-stage workflows such as cell isolation and enrichment.
Impact: Improvesthroughput during clinical development stages and supports transition toward commercial production.
Thermo Fisher Scientific Integrated CTS Platforms
Thermo Fisher’s automation-ready CTS solutions enable standardized workflows across multiple therapy pipelines.
Impact: Supports large pharmaceutical manufacturers scaling CAR-T therapy production globally.
Global Regional Analysis Including Government Initiatives and Policies Shaping the Market
North America
North America leads the robotics in cell therapy manufacturing market due to strong biotechnology infrastructure, regulatory support frameworks, and early adoption of automated manufacturing technologies. The region accounted for approximately 38.6% market share in 2025, supported by advanced research ecosystems and commercial therapy pipelines.
Government initiatives supporting advanced therapy manufacturing include:
- FDA Advanced Manufacturing Technology designation programs
- NIH funding for regenerative medicine research
- U.S. cell therapy innovation hubs
- Public-private manufacturing partnerships
These initiatives encourage adoption of robotics platforms across clinical and commercial facilities.
Europe
Europe is strengthening its position through Industry 4.0 strategies, advanced therapy regulatory pathways, and automation-driven pharmaceutical modernization initiatives.
Key policy drivers include:
- European Medicines Agency (EMA) advanced therapy frameworks
- Horizon Europe funding programs
- National robotics innovation strategies in Germany and the UK
- Cell and gene therapy manufacturing clusters in the Netherlands and France
These initiatives support distributed robotic manufacturing infrastructure across the region.
Asia-Pacific
Asia-Pacific represents the fastest-growing robotics in cell therapy manufacturing market due to increasing investments in regenerative medicine infrastructure across China, Japan, South Korea, and India.
Government initiatives include:
- Japan’s fast-track regenerative medicine approval pathway
- China’s national biotechnology development strategies
- South Korea’s cell therapy innovation funding programs
- India’s biotechnology industry development missions
These policies are accelerating adoption of automated production platforms across emerging therapy hubs.
Latin America
Latin America is gradually expanding adoption through academic research programs and regional clinical trial infrastructure development.
Government support includes:
- Public research funding initiatives
- Expansion of regenerative medicine centers
- Partnerships with international biotechnology companies
While still early-stage, robotics adoption is expected to increase with regional therapy commercialization.
Middle East and Africa
The Middle East is emerging as a strategic investment region for advanced therapy manufacturing due to healthcare diversification initiatives.
Key drivers include:
- UAE biotechnology innovation programs
- Saudi Arabia Vision 2030 healthcare transformation strategy
- Expansion of regional clinical trial ecosystems
These policies are supporting early adoption of robotic manufacturing infrastructure in advanced therapy centers.
To Get Detailed Overview, Contact Us: https://www.cervicornconsulting.com/contact-us
Read Report: Membrane Filtration Market Revenue, Trends, and Strategic Insights by 2035